 
The entropy of a system is a thermodynamic variable. Entropy is represented as $ S $, so for the irreversible process the change in entropy is $ \Delta > 0 $. it states that the entropy change of the universe increases for an irreversible or a spontaneous process. 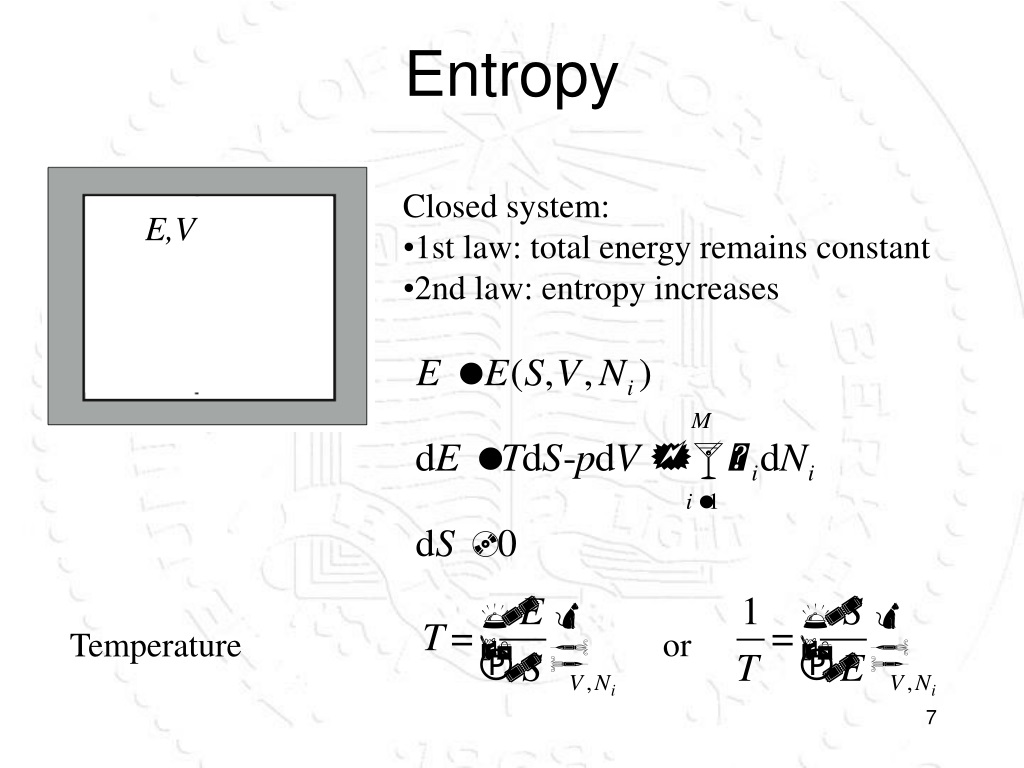
This question can also be answered by the second law of thermodynamics. And more energy level to be occupied when there is an increase in the volume of a system at constant energy. 
Because it allows higher energy levels to be significantly occupied. Increasing the energy of the system increases the entropy of that system. Also the greater the number of energy levels, the larger is the entropy. Energy always flows downhill, which causes entropy to increase. This is because entropy is a state function, the change in entropy of the system is the same, whether the process is reversible or irreversible. Entropy is related to the distribution or spread of energy among the available molecular energy levels.Īn irreversible process increases the entropy. Energy can be conserved but entropy cannot. So if Hsys is negative and Ssys is positive, what will the sign. Entropy is a non-conserved property, unlike energy. We have seen that whenever Hsys is negative (exothermic) the Ssur increases, or is positive. The entropy of a substance is lowest in the solid phase and highest in the gas phase. When a system becomes more disordered, the positions of the molecules become less predictable, and the entropy increases. The calculation of an irreversible process can be carried out by converting these irreversible processes into many reversible ones.Įntropy is defined as the property of a system that measures the degree of disorder or randomness of a system. Entropy is positive for the irreversible processes. This means it does not depend upon the path followed to change the state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
